NEWSROOM
-
Triple-negative breast cancer (TNBC), which accounts for approximately 15%-20% of all breast cancer cases, is characterized by high malignancy and a lack of effective targeted therapy. Patients with TNBC have an increased risk of distant recurrence and death within 5 years of diagnosis. Over the years, a research team from Fudan University Shanghai Cancer Center (FUSCC), led by Professor Shao Zhi-ming, the Director of Breast Surgery, and Professor Yu Ke-da, the Deputy Director of Breast Surgery, casts its insight into the regulatory mechanism that leads to the aggressiveness of TNBC. On the 10th of February, they published relevant research in the authoritative journal Nature Communications, revealing the critical role of a highly amplified gene ENSA in promoting the proliferation of TNBC. This work lays a new theoretical foundation for precision treatment strategies for TNBC.
The “culprit” of TNBC progression
What TNBC patients fear most is the rapid progression of tumors in a short period of time. Why does TNBC progress faster than other subtypes of breast cancer? What is the key “switch” that regulates the progression and deterioration of TNBC? In response to these questions that have plagued clinicians for years, Professor Shao and Professor Yu led the team to focus on the genomic abnormality of TNBC in an attempt to determine the ‘culprit’ that causes disease progression.
“Copy number alteration is a pivotal genomic event in TNBC, affecting the occurrence, development, and treatment response of tumors.” says Professor Yu Keda. Previous studies have found that TNBC has a high level of cholesterol biosynthesis, which leads to the rapid progression of tumors. This is what we usually call a regulatory "switch". However, how this "switch" makes tumor cells proliferate rapidly remains unclear, and research on relevant therapeutic value remains scarce.
Fudan researchers successfully revealed the regulatory mechanisms of the “culprit”
The research, published in Nature Communications, is titled "Copy number amplification of ENSA promotes the progression of triple-negative breast cancer via cholesterol biosynthesis".
This blockbuster research was completed in three years by a research team from FUSCC under the leadership of academic leaders, including researchers such as Chen Yi-yu, Ge Jing-yu, Zhu Si-yuan and others.
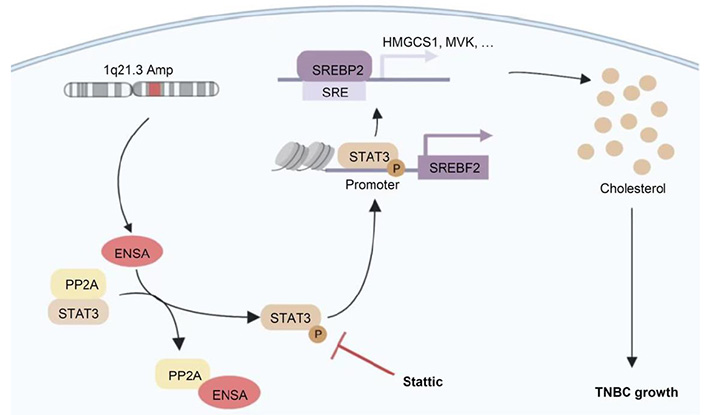
The research team first utilized the multiomics database of the world's largest TNBC cohort (FUSCC TNBC) and found that 1q, 8q, and 10p chromosome segments were commonly amplified in Chinese TNBC tumors. At the most frequently amplified 1q21.3 segment, the protein-encoding gene ENSA may be a key gene that promotes the proliferation of triple-negative breast cancer.
Professor Yu Ke-da reported that the ENSA gene was highly amplified in approximately 20% of Chinese TNBC tumors and was closely related to tumor recurrence and metastasis. After performing a series of in vitro and in vivo phenotypic studies, the research team found that interfering with ENSA expression could inhibit the proliferation of TNBC. The results of mechanistic studies revealed that ENSA could promote tumor growth by promoting the cholesterol biosynthesis program in TNBC. At the molecular level, the transcriptional activation of sterol regulatory element-binding transcription factor 2 (SREBP2) by phosphorylated STAT3 (p-STAT3) played a critical role in ENSA-induced cholesterol metabolism dysregulation. The proliferation of TNBC tumors with high ENSA expression was significantly inhibited by the STAT3 inhibitor Stattic.
"This study systematically characterizes the driving copy number alteration events in TNBC, identifies the key gene ENSA that drives TNBC tumors proliferation and cholesterol synthesis, and successfully discovers effective methods to regulate or inhibit this 'culprit'. It will provide new ideas for the subsequent precision treatment for TNBC patients." Professor Shao Zhi-ming believes that the classification of triple-negative breast cancer is becoming increasingly detailed and that the understanding of this aggressive disease is becoming clearer, which will inevitably improve the treatment of TNBC and benefit more patients.
Research results change global clinical practice
In recent years, the team led by Professor Shao Zhi-ming and Professor Yu Ke-da has always focused on various molecular subtypes of breast cancer, mainly in TNBC——"the most toxic breast cancer", and has been committed to clinical and basic translational research and has achieved a series of innovative results. Professor Shao Zhi-ming said: "The most valuable part of the output of these results is to feed back the clinical practice, practically change the clinical practice, continuously improve the efficacy and prolong the survival period of patients." A number of key achievements have been approved by the international authoritative American NCCN guidelines and American ASCO guidelines, German AGO guidelines, Chinese CBCS guidelines, etc., and have changed the global breast cancer clinical practice and made a "Chinese voice" on the global stage.
In recent years, the research team led by Professor Yu Ke-da has published many high-quality papers as corresponding authors or first authors (including coauthors) in JAMA Oncol (2020. Impact factor [IF]=32), J Clin Oncol (2020. IF=45), Nat Commun (2022. IF=15), Sci Adv (2019. IF=14), J Natl Cancer Inst (2021. IF=14), Clin Transl Med (2021. IF=11), Cancer Commun (2021. IF=10) and other international authoritative academic journals. Their papers have been cited more than 5,430 times, and the H index is 40. The achievements won the first prize of the National Maternal and Child Health Science and Technology Achievement Award in 2021, the first prize of the Science and Technology Progress Award of the China Anti-Cancer Association in 2020, the first prize of the Shanghai Science and Technology Progress Award in 2020, and the first prize of the Shanghai Anti-Cancer Science and Technology Award in 2019. Professor Yu Ke-da serves as a reviewer for the Hong Kong Research Grants Council, the Macau Science and Technology Development Foundation, the Academic Degree and Graduate Education Development Center of the Ministry of Education of China, and the National Natural Science Foundation of China. Professor Yu Keda is also the associate editor of Front Oncol breast cancer section, the editorial board member of Cancer Med, the final reviewer of Cancer Biol Med, and a member of the steering committee of the Global Early Breast Cancer Trialists Collaborative Group (EBCTCG).

